MicroTime 200 STED
- High-resolution STED microscope for super-resolution fluorescence imaging beyond the diffraction limit.
- Combines confocal single-photon counting with STED technology for nanoscale detail.
- Flexible excitation and detection channels for multi-color experiments.
- Powerful software integration for intuitive control, analysis, and automation.
- Ideal for cell biology, nanomaterials, and advanced photophysics research.
Please add items to cart and submit for quotation.
Quantity
Description
Time-resolved Confocal Fluorescence Microscope with Super-resolution Capability
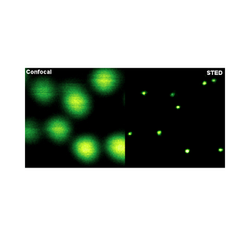
In recent years, super-resolution microscopy has gained more and more attention. It has now evolved beyond the stage of development and permits to investigate biological systems that were formerly obscured by the diffraction limit of light. One of the most popular techniques for super-resolution imaging is Stimulated Emission Depletion (STED) microscopy. STED is usually performed with confocal microscopes and is therefore ideally suited to be added to the MicroTime 200. The integration of STED into the system has been driven towards highest robustness and ease-of-use. The system permits to perform STED microscopy without lengthy alignment preparations while still having the choice to modify the system and use the full capability of the open microscopy platform MicroTime 200.
Specifications
Optical resolution :
below 50 nm (STED)
below 300 nm (confocal)
Excitation system :
Picosecond diode lasers (375 nm – 900 nm) with repetition rates up to 80 MHz inside a compact Laser Combining Unit
For STED
640 nm (excitation), 765 nm (STED laser)
Dual-species STED with 640 nm and 595 nm excitation
Dual-species STED with 640 nm and 660 nm excitation
Microscope
Inverted microscope IX 73 or IX 83 from Olympus
Specially designed right-side port for confocal microscope
The left side port and back port are still accessible (e.g., widefield imaging or TIRF)
Transmission illumination unit included
Special manual sample positioning stage with 25 mm range
Standard sample holder for 20 mm x 20 mm cover slips
Optional: epifluorescence illumination
Optional: cryostat for low-temperature measurements
Optional: combination with Atomic Force Microscope (AFM)
Objectives
Air objectives with 20x and 40x magnification (standard)
Various high-end objectives available (oil/water immersion, air spaced, IR/UV-enhanced, TIRF, or long working distance objectives)
Scanning : Piezo and Galvo scanner FLIMbee
Main optical unit
Confocal detection set-up in a compact housing with up to four parallel detection channels
Specialized high-end major dichroics with enhanced stability
All optical elements easily accessible, adjustable, and exchangeable
CCD camera for beam diagnostics and photodiode for relative power measurements
Variable beam-splitting units and exit ports to connect external devices
Detectors
Single Photon Avalanche Diodes
Hybrid-Photomultiplier Tubes
Data acquisition
Based on the method of Time-Correlated Single Photon Counting (TCSPC) in the unique Time-Tagged Time Resolved (TTTR) measurement mode
Simultaneous data acquisition of up to six channels
Software : SymPhoTime 64
Application
Stimulated Emission Depletion Microscopy (STED) / gated STED
Single Molecule Spectroscopy / Detection
Time-Resolved Fluorescence
Fluorescence Lifetime Imaging (FLIM)
Phosphorescence Lifetime Imaging (PLIM)
Fluorescence Correlation Spectroscopy (FCS)
Fluorescence Lifetime Correlation Spectroscopy (FLCS)
Foerster Resonance Energy Transfer (FRET)
Dual-focus Fluorescence Correlation Spectroscopy (2fFCS)
Scanning FCS (sFCS)
Pulsed Interleaved Excitation (PIE)
Fluorescence Anisotropy (Polarization)
Pattern Matching Analysis
Time-Resolved Photoluminescence (TRPL)
TRPL Imaging
Antibunching